BACK TO RESEARCH WITH IMPACT: FNR HIGHLIGHTS
Self-assembly is a spontaneous process where things naturally come together to form organised structures. This for example happens in our bodies, where fats come together to make a layer for cell membranes, or in technology where self-assembly helps create microchips for computers. The way these structures turn out is usually based on the properties of the starting ingredients. With the goal of being able to control the final structure, researchers are working to understand the characteristics of these starting ingredients and how they interact. This knowledge will make it possible to adjust the structures to fit specific needs, like making sensors or optical devices.

While there has been significant progress to understand the interactions of colloids – particles in the size range of nanometer – with a focus on characterising hard colloids and their interactions at interfaces, their hard character limits the use.
Understanding colloids – nanometer-sized particles
Scientists have been making progress in understanding tiny nanometer-sized particles called colloids, studying hard colloids and how they interact with surfaces. The challenge: these hard colloids have limitations in how they can be used. Researchers have been trying to use ideas from hard particles on soft microgels to create new structures with special functions.
“However, the order of the structures was always limited to a hexagonal arrangement. Even when particles were transferred from the interface to a substrate, the particles remained in a hexagonal arrangement,” explains Déborah Feller, PhD researcher at Heinrich-Heine Universität Düsseldorf

A big challenge facing researchers in this field is understanding how core-shell microgels are structured inside and how they interact with each other at the interface of air and water. Normally, hard particles are used as a model to study, but things get more complicated when it comes to soft particles. Describing what happens with soft particles, as well as in life science, becomes tricky because everything becomes more complex when dealing with soft materials.
Challenge to get the view needed for insight
Finding the right tools to measure how soft particles come together at the interface of air and water is a further challenge. Researchers need to understand not only how they interact, but also what they look like inside. The issue is that either researchers have to create their own equipment for their specific experiments or ask to use big facilities where time for measurements is very limited.

Scanning electron microscope (SEM) image of core-shell microgels with a gold core assembled on a glass substrate.
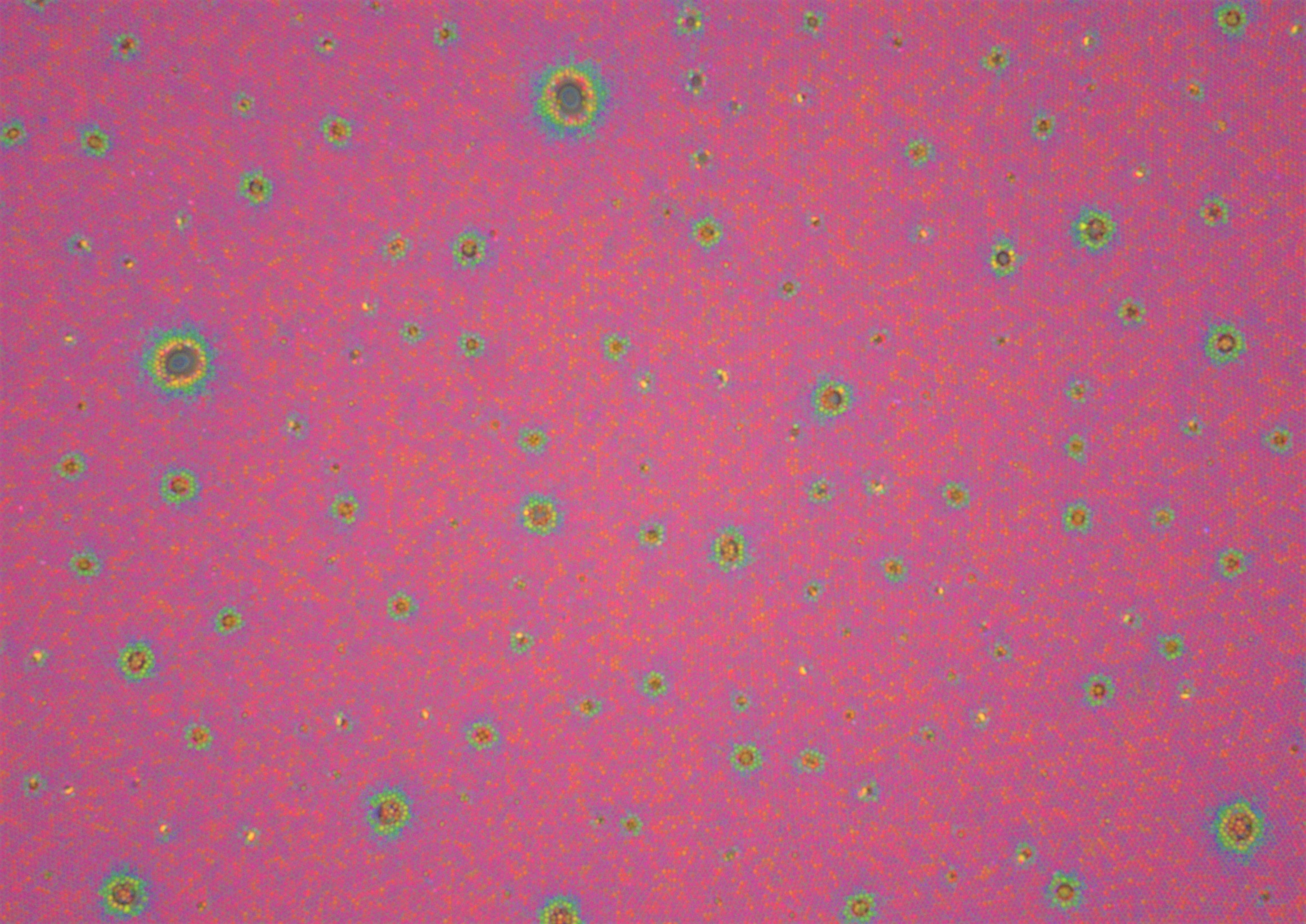
A light microscope image of core-shell microgels with a gold core assembled in a hexagonal order on a glass substrate. The cores are too small to be seen and we can only identify the microgels as dots. The particles are coated with a polymer film. The pinkish color is attributed to the optical property of the gold cores. The green/yellow spots represent some inhomogeneity/different thickness of the film. Here the light is refracted differently.
“I want to understand and control the self-assembly of core-shell microgels with a gold core at the air/water interface. I am looking at the particles at the interface or transferred to a substrate. This allows me to analyse the effects of surface area reduction on the arrangement.rnrnAdditionally, I change the final arrangement of the assembled particles on the substrate by changing the transfer process from the interface. Furthermore, I am also investigating the internal structure of core-shell microgels with a gold core in dependence of core size, shell thickness and shell softness. These different experiments (in situ and ex situ of the interface, particle characterization) should help to answer the question: How does the soft interactions affect the arrangement of the assembled particles?rn ”Déborah Feller AFR PhD researcher at Heinrich-Heine Universität Düsseldorf
A milestone: Preparing different structures from soft core-shell microgels
Déborah has been able to reach a milestone: The preparation of hexagonal and non-hexagonal (square, rectangular, centered rectangular and oblique) structures from soft core-shell microgels with a gold core on substrates.
“I investigate their optical response to light and analyse their properties after further tuning. The possibility to prepare such arrangements can encourage the investigation of the interactions of soft particles during the transfer from the interface to a substrate.”

Video of how to analyze the monolayer of core-shell particles. The glass substrate is placed under an atomic force microscope (AFM) which scans a small area of the sample to visualize the particles. The particles and their order can be seen on the screen.
Déborah Feller is a physical chemist and AFR PhD researcher at the Heinrich-Heine-Universität in Düsseldorf, supervised by Prof. Dr. Matthias Karg.
All photos provided by Déborah Feller
More about Déborah Feller
u003ch4u003eDescribing her research in one sentenceu003c/h4u003ernu003cemu003e“Synthesizing soft responsive colloids and having a look into their self-assembly at the interface to better understand soft interactions which can be transferred as model systems in life science and which can be used in technology.”u003c/emu003ernu003ch4u003eWhy she chooses her projectu003c/h4u003ernu003cemu003e“I choose the Karg research group (colloids and nanooptics) because the work is very versatile. I like the combination of synthesis, characterization and further analysis, but also building its own measurement set-up, if needed. So, I like to be interdisciplinary in chemistry and to not be focused on only one part of the aforementioned points.”u003c/emu003ernu003ch4u003eWhy she loves scienceu003c/h4u003ernu003cemu003e“I like about science, that you cannot always predict the result of your experiment and sometimes the unexpected turns out to be the most interesting result. In research, I like that a variety of single, incoherent measurements/experiments, if combined in a right way, can provide new aspects of a topic.”u003c/emu003ernu003ch4u003eWhere she sees herself in 5 yearsu003c/h4u003ernu003cemu003e“I think this question is difficult to answer as I still have two years to finish my PhD. However, I know that I want to come back to Luxembourg sooner or later and then I will have a look at the options and offers in Luxembourg to decide what I want to do.”u003c/emu003ernu003ch4u003eOn her research, peer to peeru003c/h4u003ernu003cemu003e“For my research, I am using core-shell microgels with a gold core and a polymer shell of poly(N-isopropylacrylamide). The gold cores can be overgrown in the shell and provide an optical property to the microgels, which can be seen as a peak in the UV/Vis spectrum. I will analyse the microgels before and after the overgrowth with different techniques as light, small-angle neutron and small-angle X-ray scattering to fully characterize the shell and the core. Furthermore, I am analysing monolayers of these microgels at the air/water interface under compression in a Langmuir-Blodgett trough. The analysing methods are neutron reflectometry and in situ UV/Vis spectroscopy. The monolayer will be transferred to a glass substrate during the compression to do a structural analysis with atomic force microscopy. The knowledge about the different aspects could help to control and understand the self-assembly of soft particles. This could make it possible to extend the structural variety upon hexagons, squares, rectangular etc.”u003c/emu003ernrnu003cimg class=u0022wp-image-78325u0022 src=u0022https://fnr.lu/wp-content/uploads/2024/02/Labwork_Feller-597×1024.jpgu0022 alt=u0022u0022 width=u0022311u0022 height=u0022534u0022 /u003e Déborah adding a solution of gold chloride (yellow) to a dispersion of gold core-shell microgels (pink) to overgrow the small cores in the shell. Larger cores enhance the optical properties of the particles important for their use in various applications.
Related highlights
Spotlight on Young Researchers: Virtual Reality to reconnect with your body
Body dissatisfaction affects over half of the Western population, driving the development of eating disorders, a mental illness with high…
Read more
Spotlight on Young Researchers: Lab-grown Cannabis Sativa for valuable compounds
Cannabis sativa is of great interest to scientists, with uses in areas including cosmetics, nutraceutics and pharma. Researchers are working…
Read more
Spotlight on Young Researchers: Making seawater drinkable
Water covers most of our planet, but only about 3% of it is drinkable and hard to reach for many…
Read more
Spotlight on Young Researchers: Smarter choices for complex systems
Complex systems are part of our everyday lives: smart cars, satellites, medical devices – but if they fail it can…
Read more
Spotlight on Young Researchers: Modelling Forest survival in a changing climate
Forests are dynamic ecosystems – they are used to change but it takes time to adapt. Climate change means drastic…
Read more
Spotlight on Young Researchers: Ensuring sustainable water use for Luxembourg
In Luxembourg, nearly one tenth of water consumption happens in agriculture. Changing rainfall patterns and rising irrigation needs during summer…
Read more