BACK TO RESEARCH WITH IMPACT: FNR HIGHLIGHTS
BACK TO RESEARCH WITH IMPACT: FNR HIGHLIGHTS

Divya Balakrishnan, Dipti Rani and Serena Rollo are women in science working in a field that could have a major impact on how health is managed: In the group of FNR ATTRACT Fellow César Pascual García at the Luxembourg Institute of Science and Technology (LIST), the team works on developing sensors for biochemical applications focusing on medicine.
Molecular diagnostics has seen huge advances in the last years and nanotechnology has significant potential for revolutionising medicine as well as how – and when – diseases can be detected. The NanoPH group uses nanotechnology to cater the future needs of personalised medicine, developing sensors for bio-chemical applications focused on medicine.
In the coming years, health will improve through concepts such as point of care diagnostics – earlier awareness – and personalised medicine – individually tailored treatment. The challenge: Doctors need better sensors to make the right decisions. NanoPH contributes to this goal focusing to detect the presence of the molecular biomarkers that new studies discover – as these are either found in low concentrations, or a combination of many of them is required.

The team studies two problems that are standing in the way of using sensors to implement new medical discoveries – one is managing the vast amounts of molecules that carry personal information. The second is to find a solution, a good balance, between the sensitivity and reliability of miniaturised sensors – because the more sensitive a sensor is, the more noise and variability its signal shows.
“Bio-sensors like the ones we develop use specific receptor-molecules to capture the biomarkers of interest,” the team explains, adding:
“We use electrochemical methods to manipulate and drive these receptors, and we develop new designs that improve the reliability and sensitivity of the devices. We use the same techniques as the semiconductor industry, which can speed up the implementation and scalability to detect millions of molecules for fast rapid on-line analyses.”
Below we take a closer look at the three members in César Pascual García’s international team – and which role their work plays in the group’s quest to make sensors for health a reality:

“My research may impact the future of portable devices for health monitoring”
– Serena Rollo


Serena Rollo, PhD candidate
Nationality: Italy
Serena, what do you work on as part of your PhD project and what does your work look like in practice?
“My PhD project focuses on the development of silicon based electrochemical sensors capable of being miniaturised and integrated within Lab On a Chip concepts.
“Such designs have the goal of performing on a chip what normally would be done in laboratories requiring larger infrastructures. The advantages are lower costs, less man manipulation and thus more reliability.
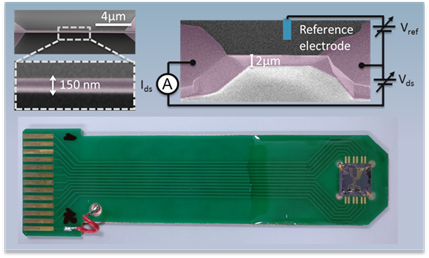
“My work includes designing, modelling and fabricating sensors starting from fundamental theory and translating the chip-prototypes into real devices using our cleanroom facilities. Then, I test them to show that they can improve the performance of current sensors.
“Using in-house models, we show the consistency of our inventions with theory, which are not only pioneering in Luxembourg, as for the first time the fabrication of a single nano-Field Effect Transistor has been made in the country, but also contribute to the state of the art of biosensors bridging the existing gap between sensitivity and reliability.
“While the sensors are currently only used as acidity sensors for a proof of concept, in the future these devices will be adapted to sense biomolecules of interest such us DNA and proteins, with the aim to contribute to the goal of better health devices.”
Why did you decide to embark on a PhD, and why did you decide to do it in Luxembourg?
“My first contact with experimental physics and the cleanroom was at my home university in Italy. I fell in love with that lab having the impression that something special was going on there. I asked the opinion of other PhD students who shared with me their experience as young researchers. At that point, I felt like challenging myself with something new, to go out of my comfort zone. I wanted to spend more time working in the cleanroom to gain more experience and acquire more knowledge and skills on micro-nanofabrication techniques.
“When I read about this project, it fulfilled the scientific expectations that I had at that time. In addition, I found an extremely multicultural environment in Luxembourg, which goes along my preferences, and I also consider is a good environment to grow. The combination of all this convinced me to embark on this experience.”
What impact do you hope your research will have on your field?
“My research may impact the future of portable devices for health monitoring. There is a gap between sensor investigations and clinical applications. Despite the fact that nanosensors could potentially detect single molecules, they are far from being commercialized, due to their poor reliability and the difficulties in measuring them.
“The needs of current applications are reliability and ease to measure while detecting the critical concentrations for the diagnosis. The geometry of the device we developed makes it more reliable, less likely to be affected by fabrication processes and easier to be measured than nano-sensors, retaining the possibility of integration within Lab on a Chip.”


“After some time working for a research institute, I wanted to become a researcher because I felt confident working in the lab and enjoyed designing and creating new things.”
– Divya Balakrishnan


Divya Balakrishnan, PhD candidate
Nationality: India
What is the focus of your work in the NanoPH group?
“My goal is making devices to help doctors detect biomarkers, which are molecules that can signal a disease or a way to cure them.
“The structure of most of these biomarkers is made of different combinations of small building blocks called nucleotides (for DNA or RNA) or amino-acids (for proteins or small fragments of them called peptides).
“The number of different combinations that can be produced is very large and the current devices used to manage them cannot cope with the variety requested by the medical applications working with these molecules. One strategy is controlling their assembly, which can be regulated with acidity.
“Our concept approach is to electrochemically produce acid in small volumes with small footprint. In the same way the microelectronics industry packages millions of devices in a small area, we miniaturise the reactors used to control the acid to increase the number of molecules that could be produced with them.
“We design and create dedicated devices where electronics can control the acidity in less than half a millimetre.
“So far, we proved several of these microreactors working simultaneously in a chip smaller than a coin of five cents and offering the largest acidity range of any electrochemical device.”
You have experience working for a research institute – why did you decide to take the plunge and become a researcher, and which parts do you particularly like?
“After some time working for a research institute, I wanted to become a researcher because I felt confident working in the lab and enjoyed designing and creating new things. I really enjoyed life in the lab and handling high-tech instruments.
“Another aspect of research that I like is sharing my work through publications or talks, which gives me a lot of satisfaction when I feel that people appreciate it. Globally it makes me think that I contribute with new things that work and have an impact. It is very interesting to work with electronics and chemistry and it rewards a lot of my scientific interests.
“Luxembourg surprised me with the numerous groups and institutions working with different expertise in broad spectrum of research activities. I thought this could provide opportunities to extend my career in different disciplines or to establish collaboration, which would favour the growth of my career.”
What drives your research and are there specific problems you want to solve?
“ ‘There is a lot of space at the bottom,’ said Richard Feynman, referring to the study of small things.
“My research deals with how small can we make chemical reactors to manage molecules. This is a passionate scientific challenge with many practical implications in pressing health topics like the request of personalised therapeutics. Reaching smaller chemical reactors and being able to pack them on a chip means that more molecules can be produced in parallel to accelerate drug discovery by exponentially increase the number of clinical trials. These devices can also find applications in other fields like environment or agriculture.”


“I learnt about nanomaterials and became fascinated by their unique properties and growing applications having direct impact on people’s lives”
– Dipti Rani

Dipti Rani, Postdoc
Nationality: India
Dipta, you work on developing sensors to detect biomarkers such as proteins or DNA molecules for disease diagnosis – your specific focus is on the surface of the sensors, can you elaborate?
“Having sensors able to detect many of these molecules in a short time is a critical tool to create new therapies. My focus is on the optimization of the surface of the sensors. The detection of biomarkers in liquids like blood or other body fluids requires the preparation of the sensor surface with particular receptors that capture specifically only the molecule with relevant medical information.
“Our challenge is to improve the simultaneous detection of multiple biomarkers to a level at which clinicians can make use of it, which requires new designs and the optimisation of all the preparation steps to provide consistent results. Doctors are demanding these kind of sensors to improve health with personalised medicines.
“To achieve all these goals, we work with concepts that upscale the fabrication of electronic devices from single sensors to a pre-industrial scale allowing the making of many devices in one process. We collaborate with immunologists to implement directly molecules currently studied in the development of cancer vaccines.”
What inspired you to embark on a career in research in this field, and why in Luxembourg?
“During my master, I learnt about nanomaterials and became fascinated by their unique properties and growing applications having direct impact on people’s lives in fields like healthcare, cosmetics, etc.
“I decided to do doctoral research, and became involved in a project studying the detection of prostate cancer, where I contributed with the development of nanosensors. I wanted to enhance my expertise in the field because these sensors can solve existing issues in cancer diagnosis and thus have a huge impact on people.
“The multidisciplinary work environment in Luxembourg involving physicists, chemists, environmental scientists from different parts of world attracted me as I could easily interact with different researchers to learn more, to collaborate and thereby enhance my career growth.”
What do you hope to achieve with your research?
“Diseases such as cancer remain undetected until late stages, because we are lagging behind with devices that can provide a reliable way of diagnosis at the right time.
“My scientific question is to answer if the materials used in microelectronics can deliver such sensors with the sensitivity, speed and reliability required at the clinical level. If yes, these sensors can improve the detection of many complex diseases because their integration with microelectronics can be used to detect simultaneously many molecules, not only for the diagnosis but also in the development of new therapies.”


About Spotlight on Young Researchers
Spotlight on Young Researchers is an FNR initiative to highlight early career researchers across the world who have a connection to Luxembourg. The campaign is now in its 4th year, with 45+ researchers already featured. Discover more young researcher stories below.
